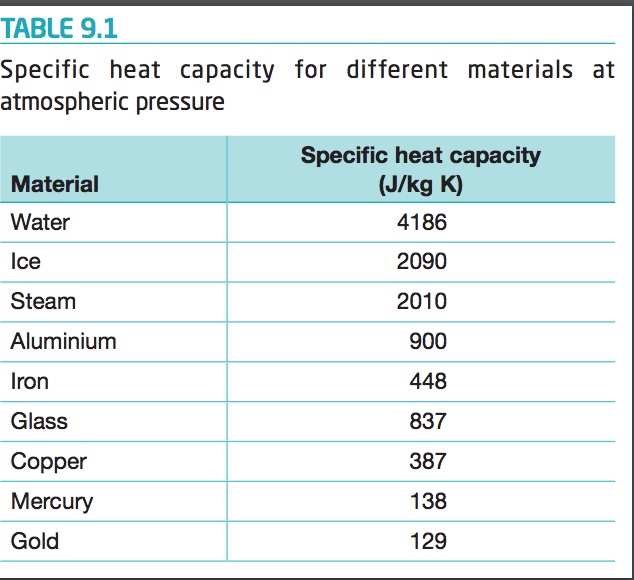
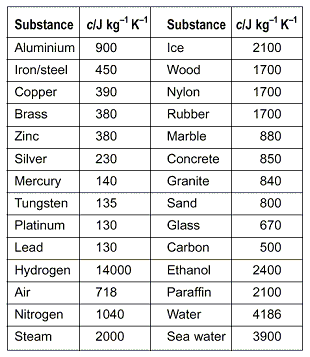
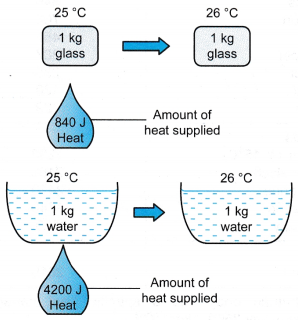
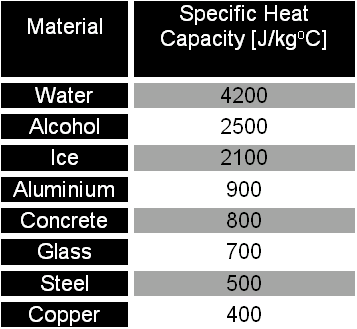
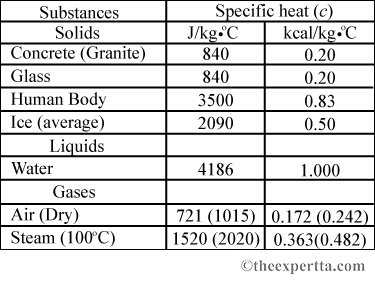
Two bars of identical mass are at 29 degrees C. One is made from glass and the other from another substance. The specific heat capacity of glass is 840 J/(kgC). When identical

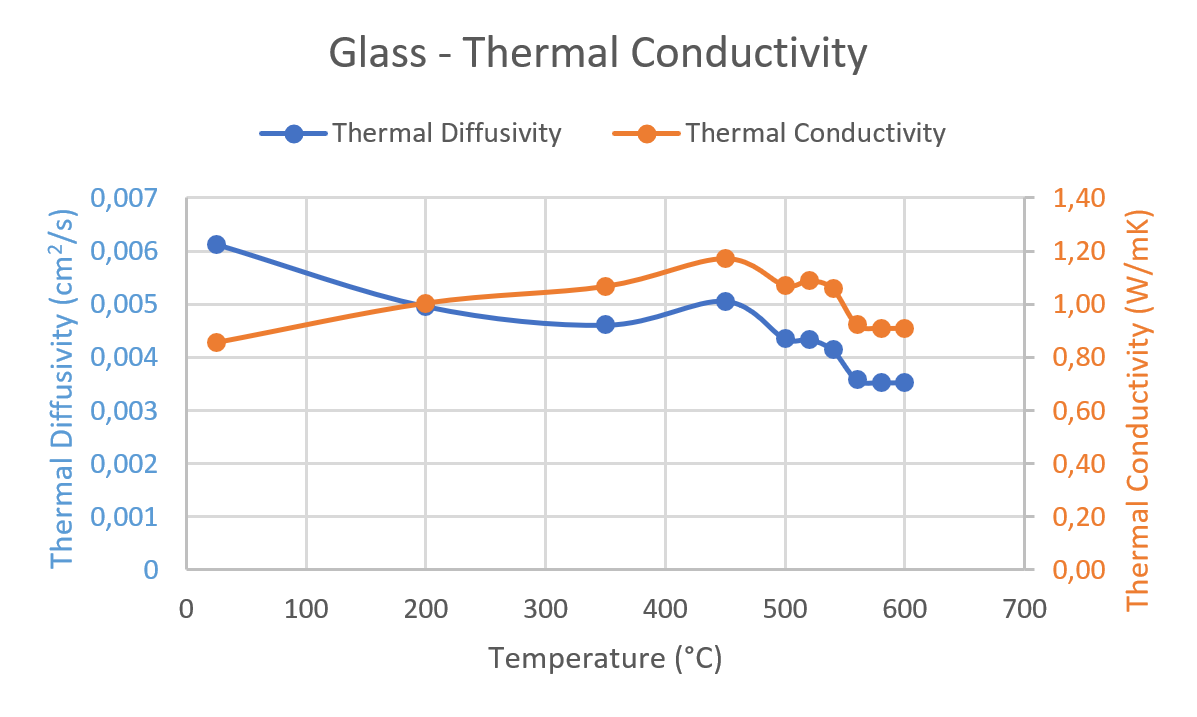
Heat capacity (C p ) vs. temperature curves in the glass transition... | Download Scientific Diagram

Specific heat and magnetization studies of spin-glass like transition in nanogranular Cu90Co10 ribbon - ScienceDirect
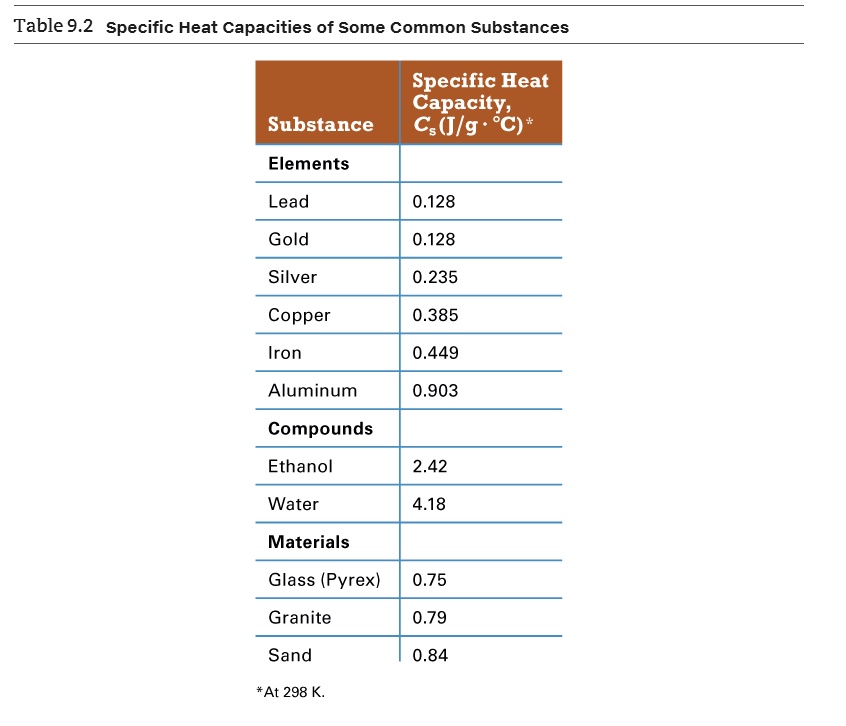
SOLVED: Table 9.2 Specific Heat Capacities of Some Common Substances Specific Heat Capacity; Cs(Jlg. *C) Substance Elements Lead 0.128 Gold 0.128 Silver 0.235 Copper 0.385 Iron 0.449 Aluminum 0.903 Compounds Ethanol 2.42

Specific heat capacities for an as-deposited thin toluene film (solid... | Download Scientific Diagram

Specific heat and magnetization studies of spin-glass like transition in nanogranular Cu90Co10 ribbon - ScienceDirect
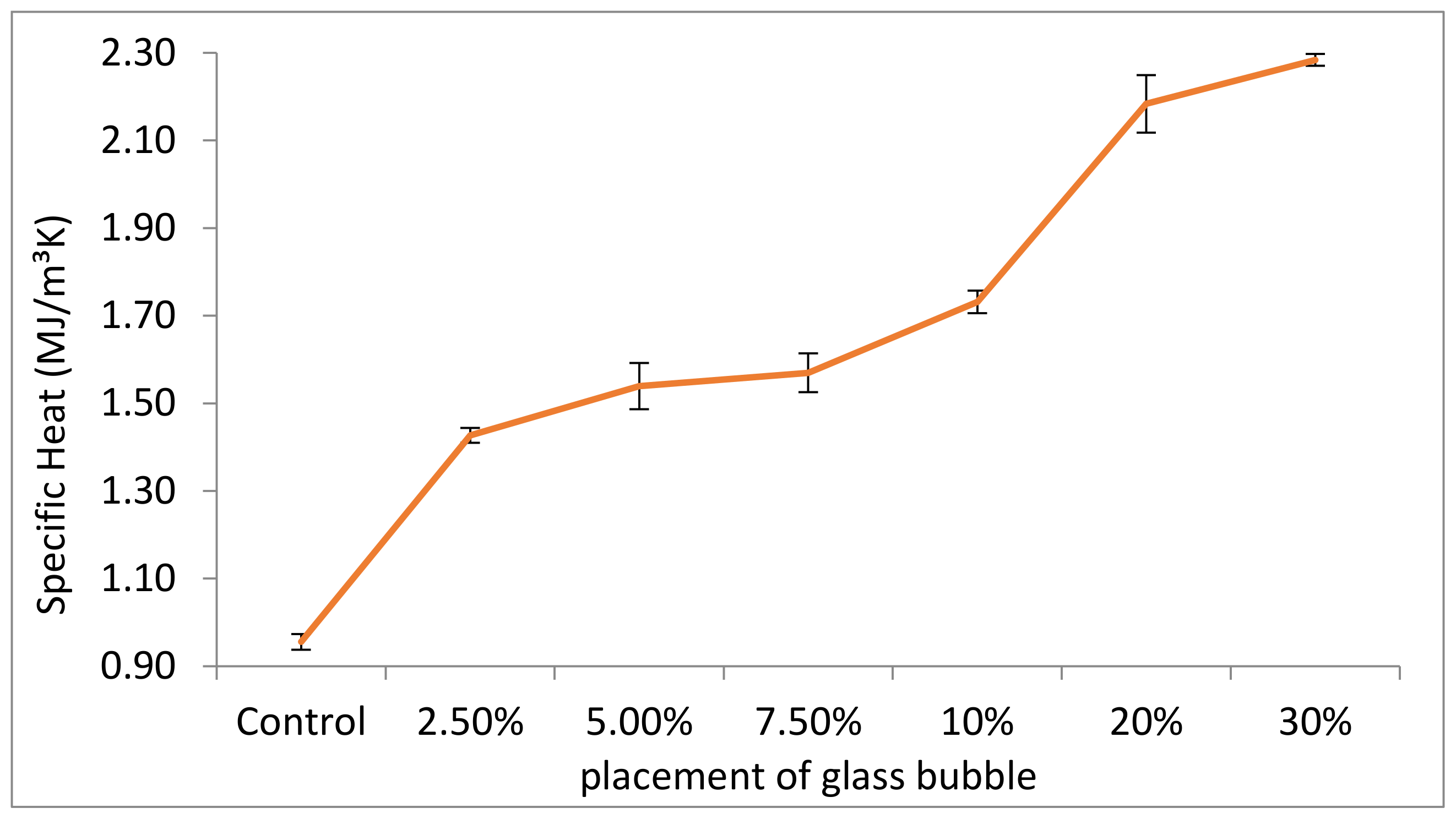
Materials | Free Full-Text | Properties of a New Insulation Material Glass Bubble in Geopolymer Concrete
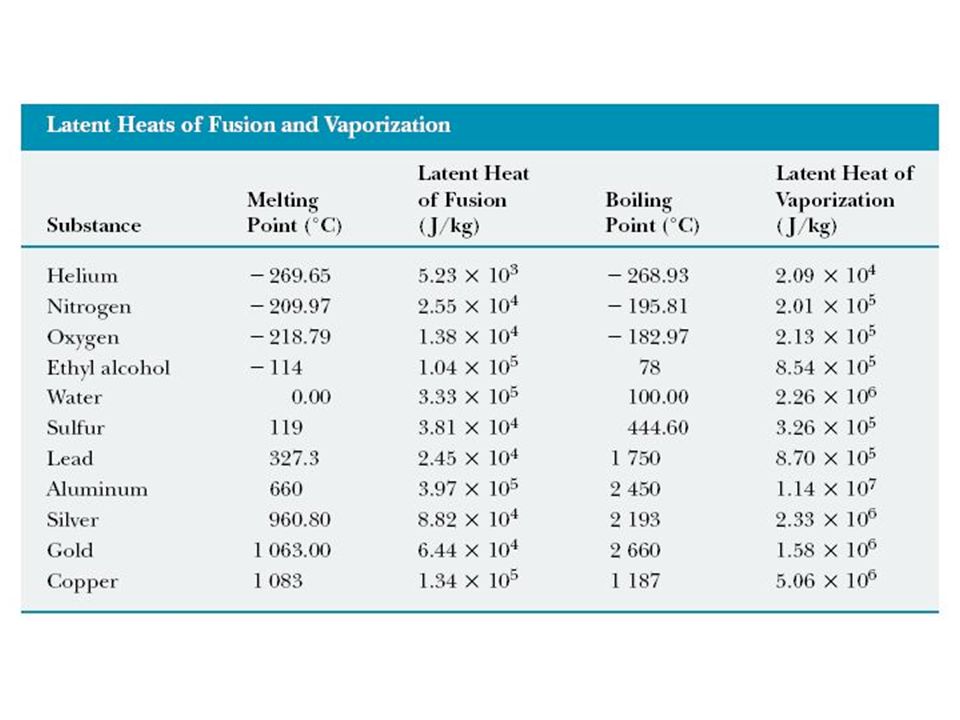
12.8 Heat and Phase Change: Latent Heat Glass of ice water at 0 C. Heat is being used to melt ice, and then only when all the ice is melted will the

Specific heat capacity Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online

SOLVED: Aheat energy of 645 J is applied to a sample of glass with a mass of 28.4g: point Its temperature increases from -11.6 'C to 15.5 'C. Calculate the specific heat


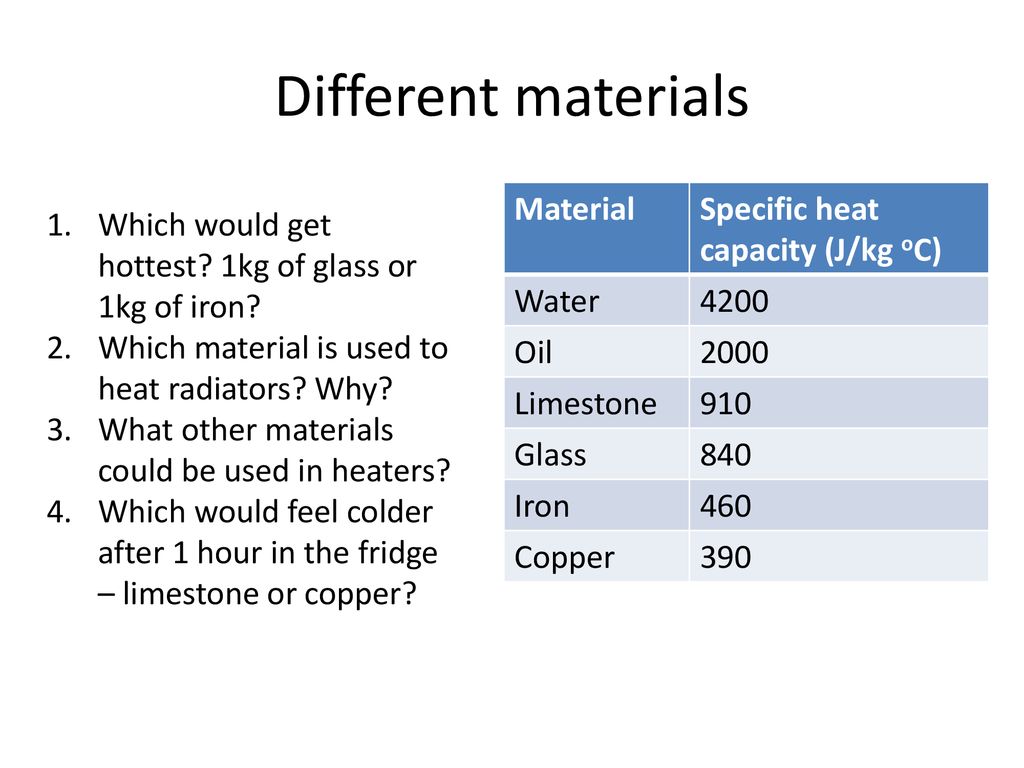

.jpg)