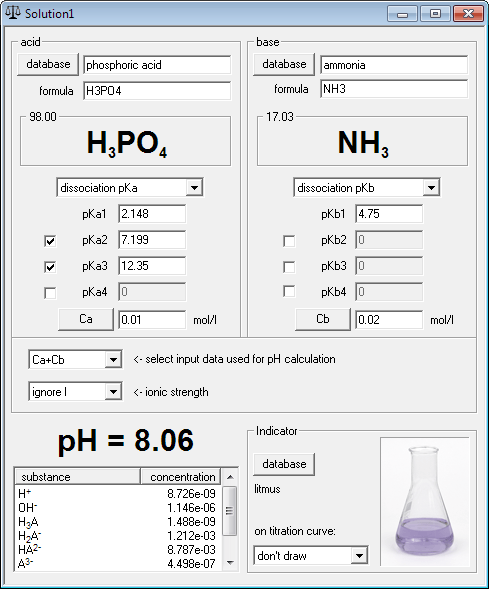
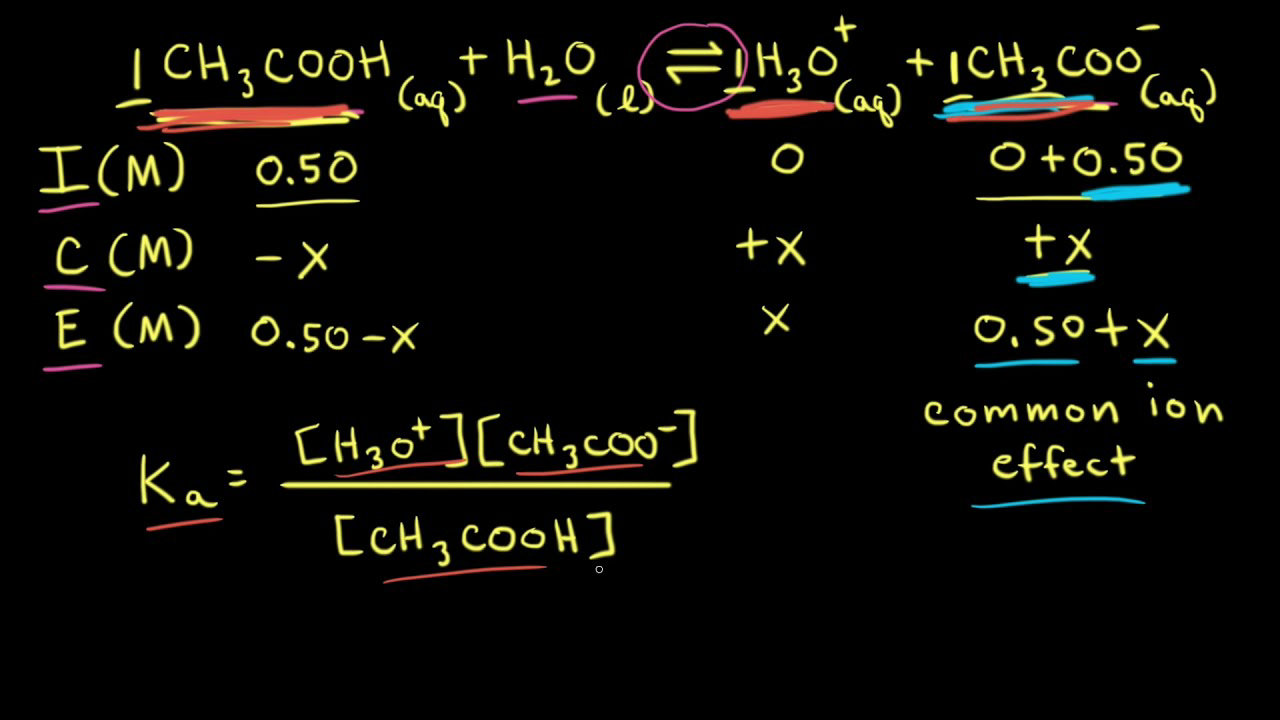Calculate the pH of a solution formed by mixing equal volumes of two solutions A and B of a strong acid having pH = 6 and pH = 4 respectively.

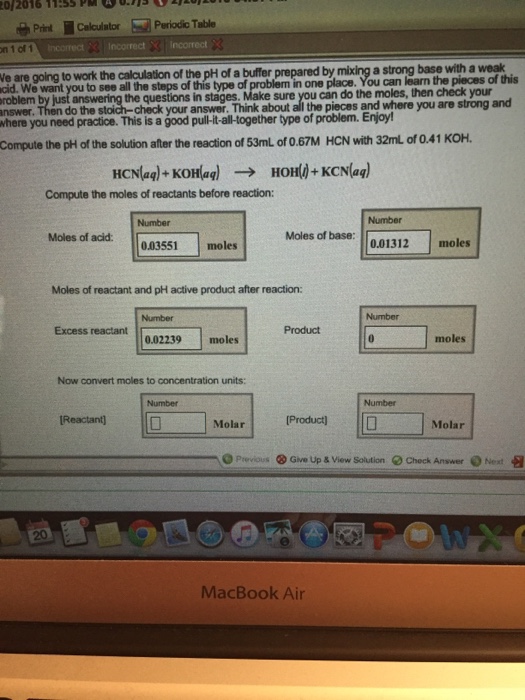
Calculate the pH of the following mixtures of strong acids, strong bases, and combination of both.a. 500 mL of 0.1 M HCl + 200 mL of 0.1 M H2SO4 + 300 mL

Calculate the pH of a solution formed by mixing equal volumes of two solutions A and B of a strong acid having pH = 6 and pH = 4 respectively.