Antibodies | Free Full-Text | Current Advancements in Addressing Key Challenges of Therapeutic Antibody Design, Manufacture, and Formulation

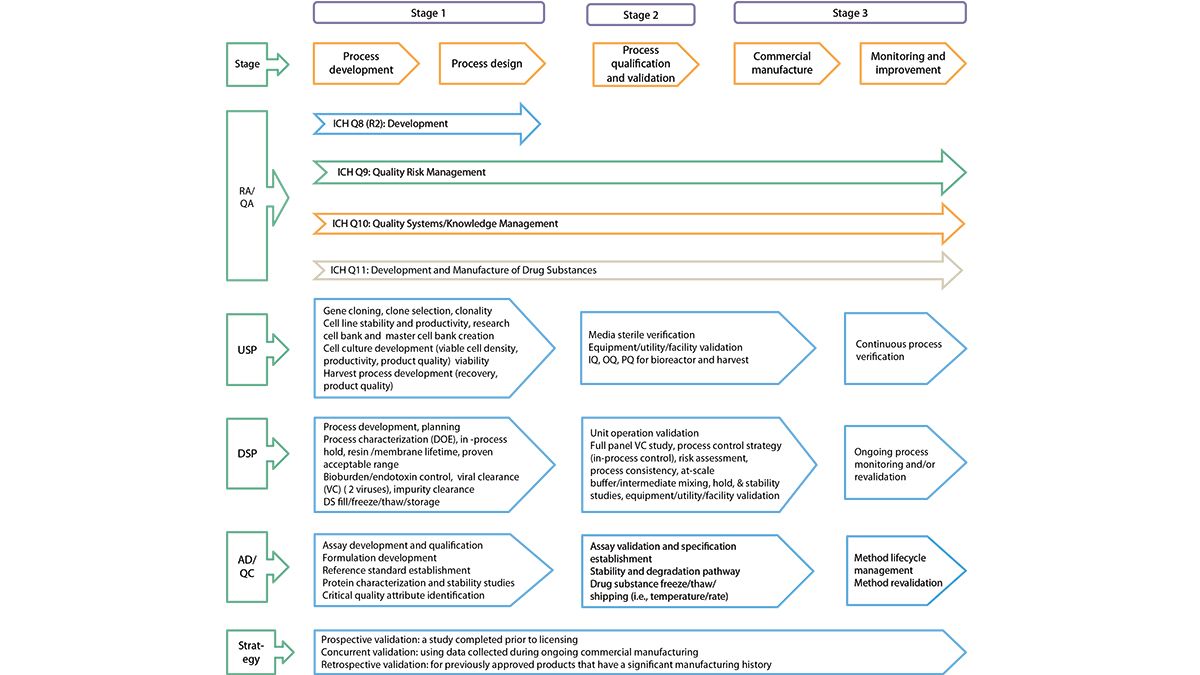
Quality By Design for Monoclonal Antibodies, Part 1: Establishing the Foundations for Process Development - BioProcess InternationalBioProcess International

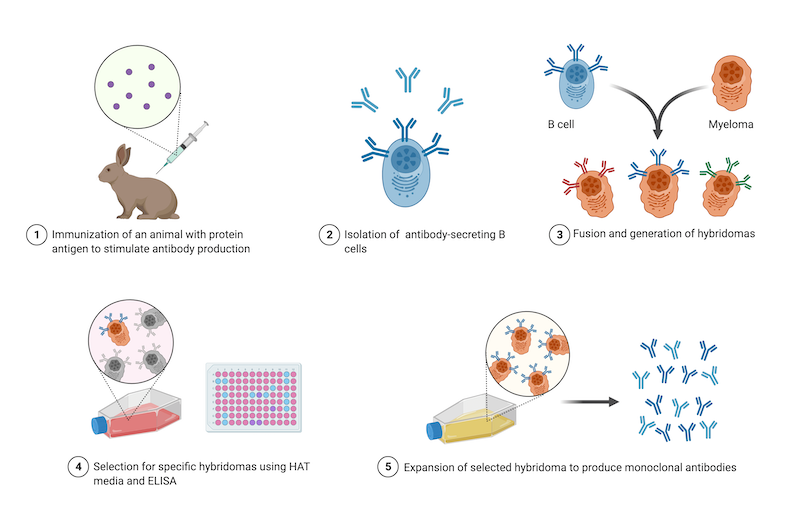
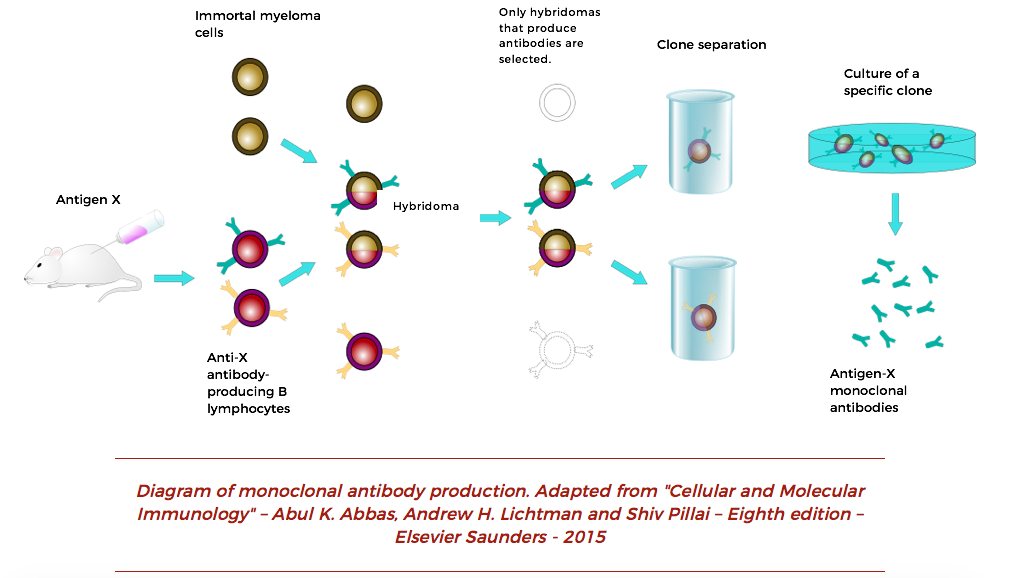
Monoclonal antibodies: Diagnostics Discovery Laboratory: University of Nevada, Reno School of Medicine

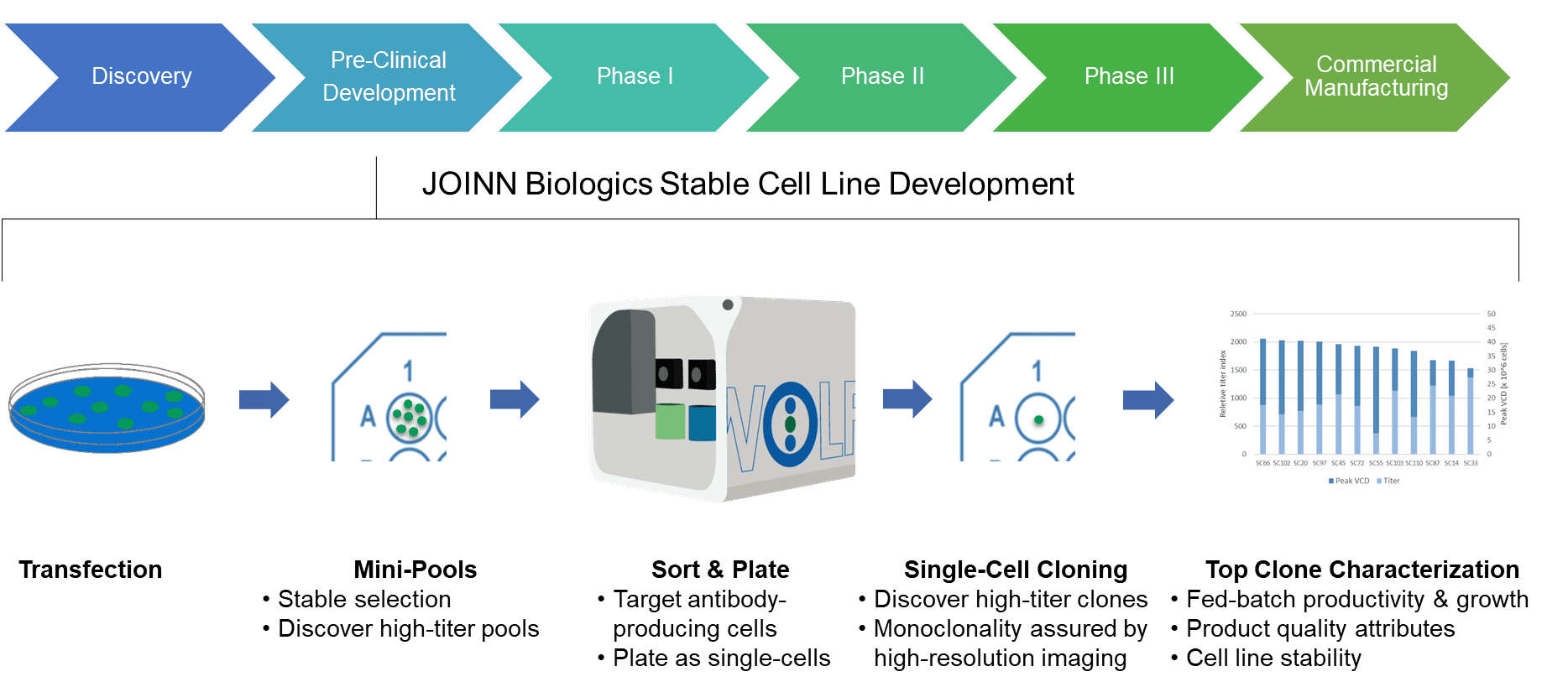
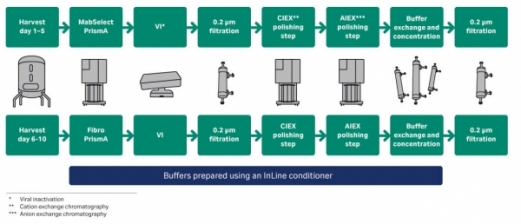
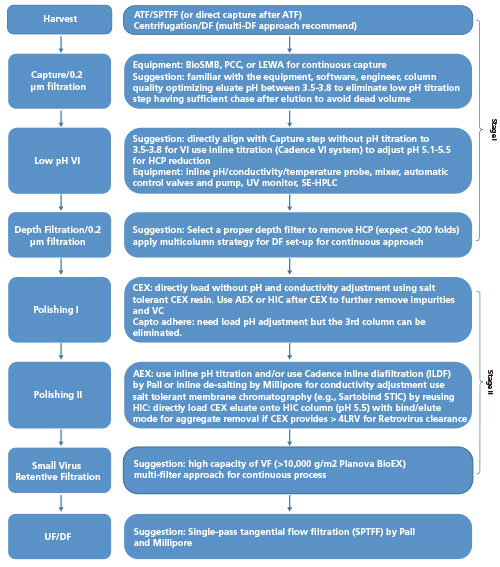
Transfer, Implementation and Late Stage Development of an End-To-End Single-Use Process for Monoclonal Antibody Manufacture | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
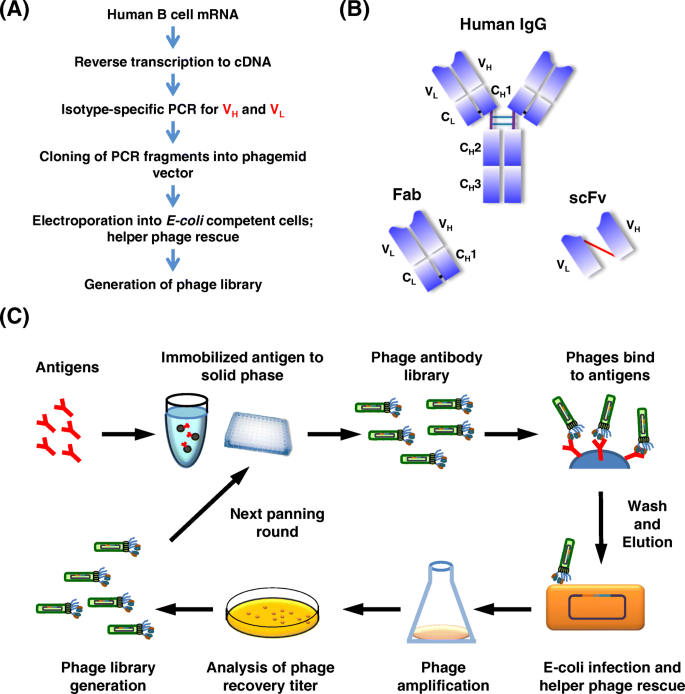
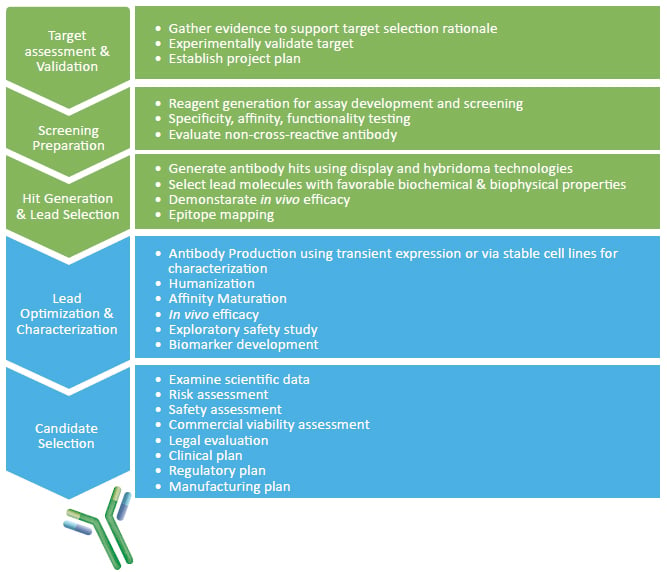
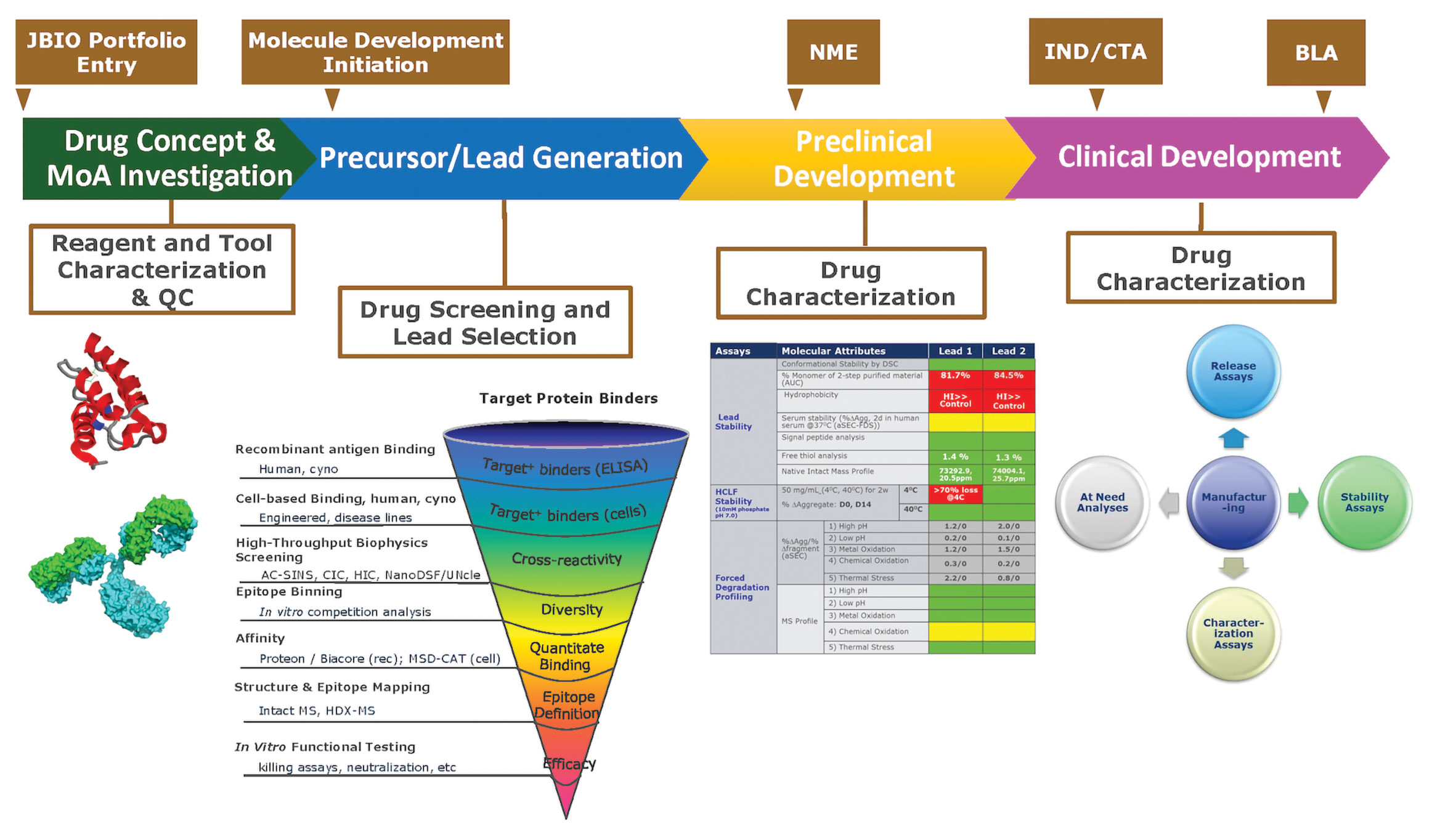
Development of therapeutic antibodies for the treatment of diseases | Journal of Biomedical Science | Full Text

Decision-Support Tools for Monoclonal Antibody and Cell Therapy Bioprocessing: Current Landscape and Development Opportunities - BioProcess InternationalBioProcess International